Visual EM
A Case Report of Acute Appendicitis Complicated by Appendicoliths
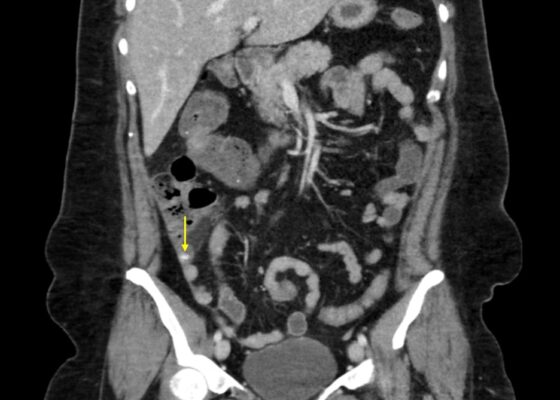
DOI: https://doi.org/10.5070/M5.52213Contrast-enhanced CT of the abdomen and pelvis was obtained. Coronal CT demonstrated a dilated appendix measuring up to 1.2 cm in diameter with multiple appendicoliths (yellow arrows), the largest measuring 1.1 cm. Mild periappendiceal fat stranding was present, consistent with acute appendicitis complicated by appendicoliths. The surrounding bowel and colon were normal in caliber and distribution. An additional coronal view demonstrated a dilated appendix (yellow arrow) containing an appendicolith with periappendiceal inflammatory changes. No evidence of perforation, abscess, or drainable fluid collection was identified.
Case Report: Acute Dyspnea in a Young Female
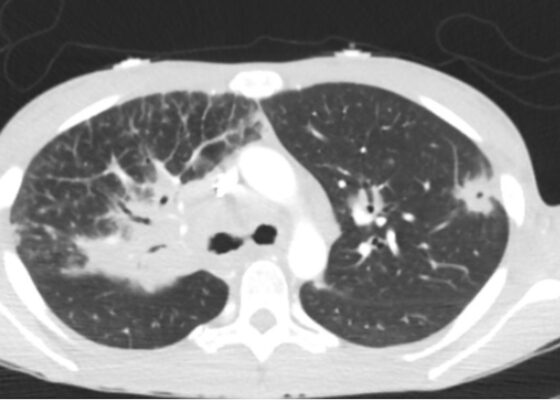
DOI: https://doi.org/10.5070/M5.52254The patient underwent computer tomography (CT) imaging of the chest, abdomen, and pelvis. Computer tomography of the chest showed bilateral diffuse pulmonary nodules in a perilymphatic distribution (yellow arrows), and diffusely increased mediastinal and hilar soft tissue densities (blue circle), likely representing lymphadenopathy; a left upper lobe lesion with central cavitation (orange circle) was also seen likely to be associated with the same disease process as the extensive lymphadenopathy. Imaging of the abdomen and pelvis was significant for diffuse hypodensities of the liver and spleen suggesting a multi-system process.
The Rash That Didn’t Blanch: A Case Report of Adult-Onset IgA Vasculitis with Underlying Cirrhosis and IgA Nephropathy
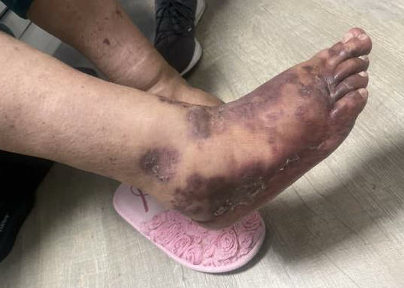
DOI: https://doi.org/10.5070/M5.52253Given the patient’s physical exam findings of tender palpable purpura to the lower extremities, dermatology was consulted for suspicion of acute small vessel vasculitis. A punch biopsy for hematoxylin and eosin (H&E) stain and direct immunofluorescence (DIF) was subsequently performed. The H&E demonstrated nonspecific purpura.
A Case Report on an Open Fracture Dislocation Injury of the Proximal Phalanx of the Thumb Resulting from Playing Cricket
DOI: https://doi.org/10.5070/M5.52278There was an open injury to the volar aspect of the right thumb at the interphalangeal joint, with exposed bone. There was no active bleeding from the wound. He had intact sensation to the entire thumb and hand. His radial pulse was normal with normal capillary refill in all digits of his right hand. He had intact wrist flexion, extension, abduction, and adduction; however, he was unable to flex or extend his thumb secondary to the injury. He had no other injuries to the rest of his right upper extremity. An x-ray was obtained which showed a right thumb proximal phalanx intra-articular fracture (proximal fracture fragment outlined in yellow, distal fracture fragment outlined in pink) at the interphalangeal joint with dorsal dislocation of the distal phalanx (outlined in red). There were no radiopaque foreign bodies.
Woman with a Blackened Tongue: A Case Report
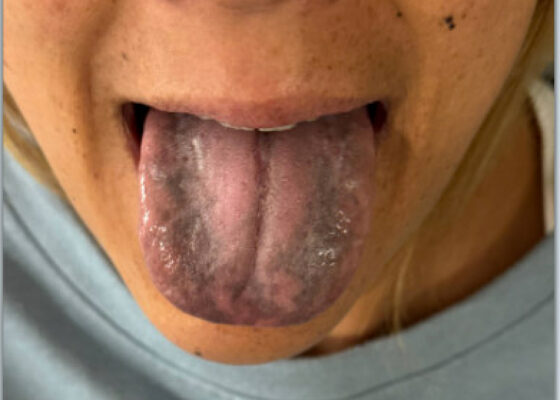
DOI: https://doi.org/10.5070/M5.52329The patient’s vital signs were blood pressure 109/72 mmHg, respiratory rate 18 breaths per minute, pulse 93 beats per minute, temperature 97°F. Physical examination revealed notable black discoloration of the tongue and gingiva, along with hyperpigmented skin exhibiting numerous diffuse nevi distributed across the face and upper extremities.
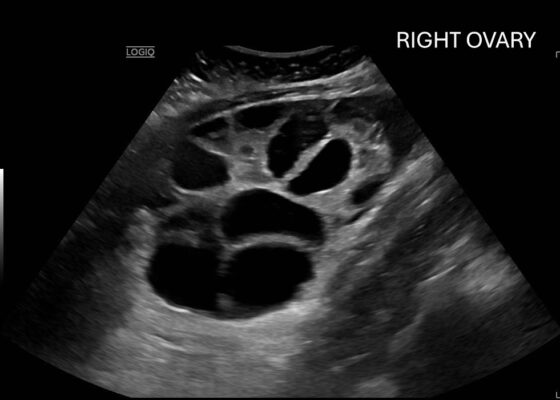
A Woman’s Infertility Journey Complicated by Severe Ovarian Hyperstimulation Syndrome – A Case Report
DOI: https://doi.org/10.5070/M5.52309Computed tomography (CT) of the abdomen and pelvis demonstrated pleural effusions in the lung bases (blue stars), ascites (blue stars), and enlarged ovaries with multiple cysts/follicles (white arrows). A formal pelvic US also demonstrated large volume ascites (white diamonds) and bilateral ovarian enlargement with numerous cysts. A CT angiogram (CTA) of the chest demonstrated small bilateral pleural effusions and no obvious pulmonary embolism.
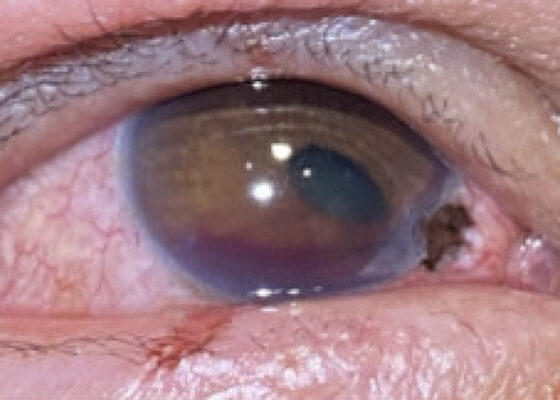
A Case Report of Corneoscleral Laceration with Open Globe Injury and Iris Prolapse
DOI: https://doi.org/10.5070/M5.52326Closer examination of the right eye revealed a 2 mm x 1 mm corneoscleral laceration to the 4-5 o'clock position at the border of zone I (yellow highlighting) and II (blue highlighting) with iris prolapse (yellow star) and plugging. Additionally, the affected eye revealed a peaked pupil (cyan highlighting) in the inferonasal direction with associated corectopia and a 2 mm Grade I hyphema (pink highlighting). Immediately inferior to the corneal laceration, 1 mm epithelial injury of the lower palpebra was present (green cross). Fluorescein exam demonstrated uptake at the 4-5 o'clock position (yellow arrow) but was negative for Seidel’s sign.
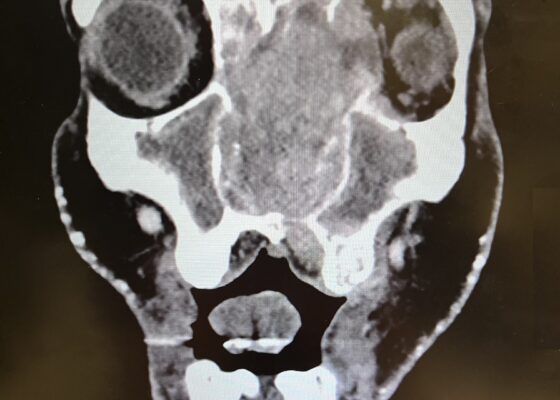
Diagnosis of Sinonasal Carcinoma in the Emergency Department: A Case Report Highlighting Red Flag Symptoms
DOI: https://doi.org/10.5070/M5.52257Physical exam revealed a gross deformity of the left side of the face with soft tissue swelling with no overlying skin changes. The left eye was proptotic and completely immobile, with cranial nerve (CN) 3, 4, and 6 palsies. Additionally, a large obstructive mass was noted in the left naris with resultant rightward displacement of the nasal septum. Intraocular pressures (IOPs) were measured to be 6 mmHg in the right eye and 11 mmHg in the left eye.
Imaging, including maxillofacial and neck soft tissue CTs revealed a large, destructive, soft tissue mass centered in the nasal cavity with significant osseous destruction of the midface and skull base (red highlighted area). There was mild intracranial extension with the mass abutting or infiltrating the inferior frontal lobes (green highlighted area). The nasal cavity and nasopharynx were obstructed (orange highlighted area). The mass invaded the left orbit with associated left-sided proptosis, left globe deformity, and compressed the left optic nerve and the left optic chiasm (blue arrows).