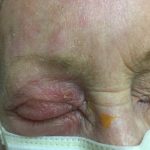
Herpes Zoster Ophthalmicus
History of present illness:
A 75-year-old woman receiving chemotherapy for metastatic breast cancer presented to the emergency department for three days of right eye pain and redness. She noted a rash above the affected eye accompanied by a headache in the same region, which she described as feeling that she had been “punched in the face.” Vision in both eyes remained intact. She denied the presence of scotoma, photopsia, or metamorphopsia.
Significant findings:
Physical exam was significant for an erythematous maculopapular rash in the right ophthalmic nerve (V1) region, a crusted vesicle at the right upper eyelid, and surrounding eyelid edema. Visual acuity remained at baseline and intraocular pressure was within normal limits. External slit lamp exam with fluorescein staining was remarkable for pseudodendrites in the inferior-temporal cornea approximately 1.5 mm from the limbus. Ophthalmology was consulted and completed an in-depth evaluation of the corneal lesions. They found no evidence of anterior chamber reaction and performed a complete dilated fundus examination which demonstrated no retinal involvement. The patient was then discharged on acyclovir and erythromycin ointment with close follow-up.
Discussion:
Herpes zoster ophthalmicus occurs due to reactivation of the varicella zoster virus within the first division of the trigeminal nerve. Elderly and immunocompromised are at significantly increased risk of contracting herpes zoster ophthalmicus.1 Prodromal symptoms include headache, malaise, and myalgias. Patients commonly develop a unilateral maculopapular rash in a single dermatome that progresses into vesicles then pustules. Patients with skin lesions involving the nasociliary dermatome (Hutchinson’s sign) should be considered at high risk for ocular involvement.2
Acyclovir, valacyclovir, and famciclovir are approved by the Food and Drug Administration (FDA) for the treatment of herpes zoster. Oral acyclovir (800mg five times daily) administered within 72 hours of onset has been shown to significantly reduce pain, the severity of the rash, and the incidence of late ocular inflammatory complications and postherpetic neuralgia.3,4 There is some evidence that supports the use of steroids for pain reduction and accelerated healing.5,6 However, due to side effects and potential complications, steroids should only be considered as a supplementary treatment to antiviral therapy.7 Referral to ophthalmology is crucial. Left untreated, eye complications including neurotrophic keratopathy, oculomotor palsies, glaucoma, optic neuritis, and acute retinal necrosis may result in permanent vision loss.8
Topics:
Herpes zoster ophthalmicus, ophthalmology, slit lamp exam, rash.
References:
- Gnann JW, Whitley RJ. Clinical practice. Herpes zoster. N Engl J Med. 2002;347(5):340–346. doi: 10.1056/NEJMcp013211
- Zaal MJ, Volker-Dieben HJ, D’Amaro J. Prognostic value of Hutchinson’s sign in acute herpes zoster ophthalmicus. Graefes Arch Clin Exp Ophthalmol.2003;241(3):187-191. doi: 10.1007/s00417-002-0609-1
- McKendrick MW, McGill JI, White JE, Wood MJ. Oral acyclovir in acute herpes zoster. Br Med J Clin Res Ed.1986;293(6561):1529.
- Hoang-Xuan T, Buchi ER, Herbort CP, et al. Oral acyclovir for herpes zoster ophthalmicus. 1992;99(7):1062-1070; discussion 1070-1071. doi: 10.1016/S0161-6420(92)31849-4
- Wood MJ, Johnson RW, McKendrick MW, et al. A randomized trial of acyclovir for 7 days or 21 days with and without prednisolone for treatment of acute herpes zoster. N Engl J Med.1994; 330:896–900. doi: 10.1056/NEJM199403313301304
- Whitley RJ, Weiss H, Gnann JW, et al. Acyclovir with and without prednisone for the treatment of herpes zoster. A randomized, placebo-controlled trial. The National Institute of Allergy and Infectious Diseases Collaborative Antiviral Study Group. Ann Intern Med. 1996;125(5):376–383. doi: 10.7326/0003-4819-125-5-199609010-00004
- Cohen JI. Herpes Zoster. N Engl J Med.2013;369:255-263. doi: 10.1056/NEJMcp1302674
- Liesegang TJ. Herpes zoster ophthalmicus natural history, risk factors, clinical presentation, and morbidity. 2008;115(2):S3-S12. doi: 10.1016/j.ophtha.2007.10.009