CT
A Case Report of Hydropic Gallbladder Presenting as Right Lower Quadrant Abdominal Pain
DOI: https://doi.org/10.21980/J8DD26Computed tomography (CT) of the abdomen and pelvis with contrast was ordered, and general surgery was consulted for the initial working diagnosis of acute appendicitis. However, the CT scan resulted with findings of a markedly distended gallbladder measuring approximately 14.5 x 4 centimeters (cm) with marked gallbladder wall thickening (magenta) and pericholecystic fat stranding (cyan). The appendix was not dilated and had no inflammatory changes or edema. Follow-up right upper quadrant ultrasound confirmed the diagnosis of acute cholecystitis.
A Case Report of Right Atrial Thrombosis Complicated by Multiple Pulmonary Emboli: POCUS For the Win!
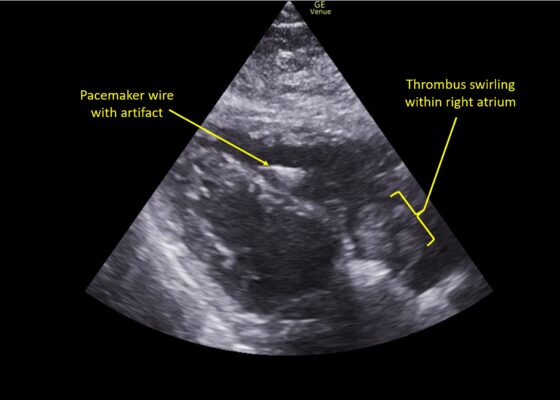
DOI: https://doi.org/10.21980/J8TM07Pulmonary POCUS was performed by the ED physician (GE Venue, C1-5-RS 5MHz curvilinear transducer), and lung examination was unremarkable with no pleural effusion, pneumothorax, or infiltrate. Subxiphoid views (GE Venue, 3Sc-RS 4MHz phased-array transducer) were obtained because this patient’s COPD with severe pulmonary hyperexpansion made parasternal and apical 4-chamber views suboptimal. A large thrombus can be seen within the right atrium (movie 1, images 1, 2). This has a serpiginous, rounded appearance and is mobile, appearing to swirl within the right atrium with intermittent extrusion through the tricuspid valve. A pacemaker wire is also visible within the right ventricle as a non-moving, hyperechoic, linear structure with posterior enhancement artifact. Pericardial effusion is not present.
Retropharyngeal Abscess in an Adult Patient Presenting with Neck Fullness and Dysphagia: A Case Report
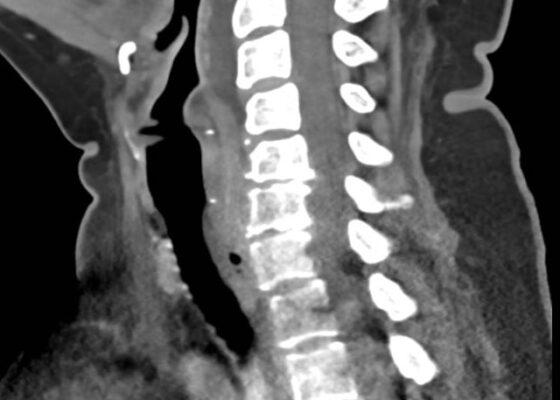
DOI: https://doi.org/10.21980/J8M36GContrast-enhanced CT soft tissue of the neck showed evidence of a prevertebral/retropharyngeal fluid collection, extending from the odontoid tip to the inferior C4 vertebral body margin, measuring 5.4 x 1.0 x 3.3 centimeters (cm) in size (yellow lines) without gross airway narrowing.
A Man With Chest Pain After An Assault – A Case Report
DOI: https://doi.org/10.21980/J8J93SOn exam, we found a suspected chest wall abscess with surrounding erythema (blue arrow). The patient underwent CT of the chest which showed a comminuted displaced midsternal fracture (yellow arrow) with moderate fluid and air anteriorly (red arrow), consistent with an abscess. His laboratory results had no significant abnormalities.
A Case Report Evaluating Gastric Emphysema versus Emphysematous Gastritis
DOI: https://doi.org/10.21980/J8ZH26A CT scan of the abdomen and pelvis was obtained and revealed gas within the gastric wall at the fundus (blue arrows), concerning for gastric emphysema versus emphysematous gastritis. There was no gastric wall thickening, free air, bowel obstruction, drainable fluid collection, or evidence of portal venous gas. Incidentally, hepatomegaly and likely hepatic steatosis were also noted.
Telescoping into Adulthood: A Case Report of Intussusception in an Adult Patient
DOI: https://doi.org/10.21980/J8Q06CComputed tomography imaging of the abdomen and pelvis with intravenous and oral contrasts was obtained. In the axial view, one will see a concentric ring formed by layers of bowel, mesenteric vessels, and fat (red arrow and circle); this is the equivalent of the ultrasonographic “target sign.” The inner ring (blue arrow) represents the lead point causing telescoping of the bowel. One can see that the proximal bowel is dilated (yellow arrow). In the coronal view, one can see an obstructive mass, also known as the lead point (red arrow), located in the lumen of the descending colon. Located proximal to the lead point are dilated loops of bowel with edematous changes and fat stranding (pink circle). The proximal portion of the bowel will take on a concentric appearance with the telescoping loop of bowel.
Trauma by Couch: A Case Report of a Massive Traumatic Retroperitoneal Hematoma
DOI: https://doi.org/10.21980/J84D2QUpon arrival at the trauma center, a FAST revealed a large, well-circumscribed abnormality (red outline) deep to the liver (blue outline and star) and gallbladder (green outline and star). The right kidney and hepatorenal space were not clearly visualized. The remainder of the FAST showed no free fluid in the splenorenal space, pelvis, and no pericardial effusion. He had lung sliding bilaterally.
A Patient with Generalized Weakness – A Case Report
DOI: https://doi.org/10.21980/J8593CThe CT of the abdomen and pelvis showed evidence of a large subcapsular rim-enhancing fluid collection with multiple gas and air-fluid levels along the right kidney measuring 8 x 4 cm axially and 11 cm craniocaudally (blue outline) with mass effect on the right renal parenchyma (yellow outline). Another suspected fluid collection adjacent to the upper pole of the right kidney measuring 4 x 3.4 cm was noted (red outline). Bilateral pyelonephritis was suggested without hydronephrosis or nephrolithiasis. The findings suggested complicated pyelonephritis with emphysematous abscess and hematoma formation.